Auteur : Dylan Daniel, PhD, directeur au développement scientifique
Date: November 2017
ID8-Luc est un nouveau modèle murin syngénique développé pour traiter le cancer orthotopique des ovaires. Le cancer de l'ovaire demeure un besoin médical non satisfait important, avec un taux de survie à 5 ans de 17 % chez les patientes chez qui on a diagnostiqué un cancer de l'ovaire de stade IV très avancé.
Le cancer de l'ovaire est un besoin médical non satisfait et particulièrement mortel car il n'est souvent détecté que lorsque la maladie est assez avancée. En 2017, environ 22 440 femmes seront diagnostiquées d'un cancer de l'ovaire aux États-Unis, et environ 14 080 décès auront lieu. La chimiothérapie combinée à base de platine et de taxane antimitotique est la norme de soins aux États-Unis, mais si ces options de traitement sont souvent efficaces au départ, les rechutes et la progression sont fréquentes. De nouvelles approches thérapeutiques sont nécessaires pour traiter le cancer de l'ovaire.
En se basant sur les récents succès de l'utilisation du système immunitaire pour traiter d'autres cancers, les scientifiques spécialisés dans la découverte de médicaments ont exploré des approches d'immunothérapie dans le cancer de l'ovaire. Il y a actuellement plus de 50 essais cliniques testant des approches et des combinaisons d'immunothérapie dans le cancer de l'ovaire ; cependant, il existe un nombre limité de modèles de cancer de l'ovaire chez des souris syngéniques pour permettre une pharmacologie préclinique. Labcorp has developed the ID8 syngeneic mouse cell line model for testing standard therapeutics as well as for immuno-oncology applications. L'ID8 est dérivé de cellules épithéliales de surface ovarienne de souris C57BL/6 qui ont été transformées par passage sérié in vitro 1. La luciférase a été exprimée dans les cellules ID8 (ID8-Luc-mCherry-Puro) pour permettre le suivi de la croissance orthotopique (intrapéritonéale) des tumeurs par imagerie en bioluminescence (BLI).
La cinétique moyenne de croissance des tumeurs et la cinétique de croissance individuelle d'ID8-Luc-mCh-Puro, telles que surveillées par BLI, sont illustrées par les figures 1A et 1B respectivement. L'implantation de 1,0×107 cellules par souris a donné une croissance tumorale plus régulière que celle de 5,0×{106 cellules par souris chez les souris albinos C57BL/6. Comme prévu, le signal (photons/sec) est plus élevé chez les souris albinos C57BL/6 (ligne verte, figure 1B, groupe 2) que chez les souris sauvages C57BL/6 (ligne rouge, figure 1B) en raison d'un manque de pigment de mélanine dans leur peau qui absorbe la lumière émise par les cellules tumorales. La figure 1C montre des images BLI représentatives de la progression de la maladie orthotopique ID8-Luc-mCh-Puro.
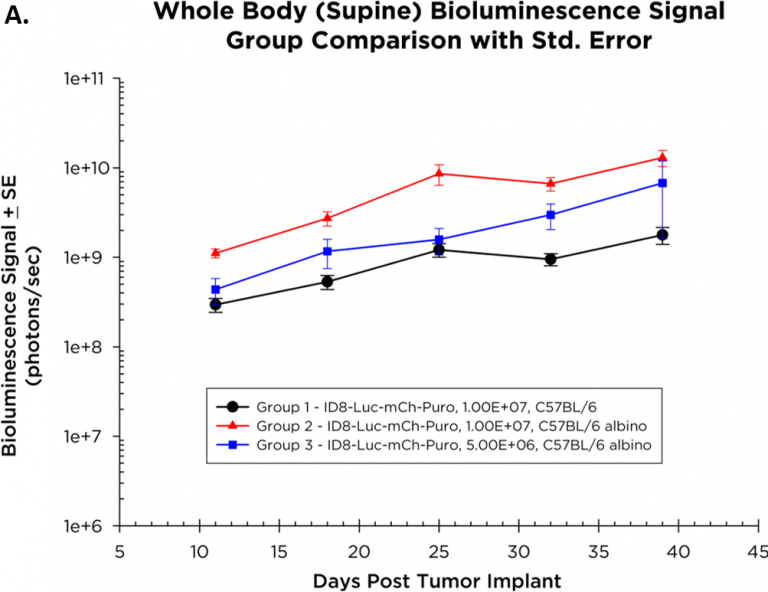
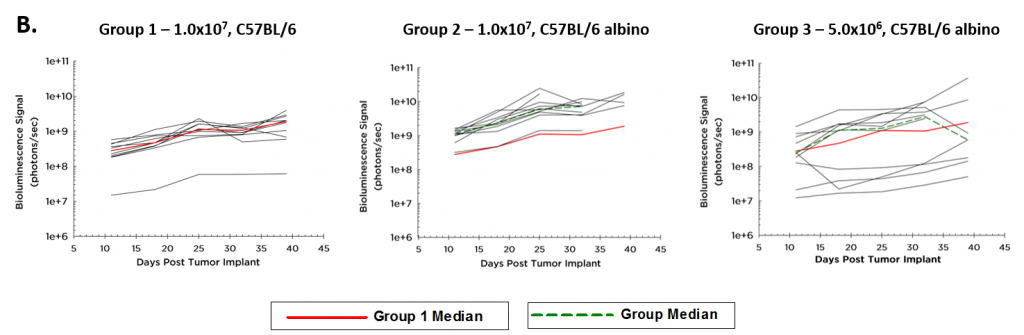
Fig. 1B – Growth Kinetics of Tumor-ID8-Luc-mCh-Puro
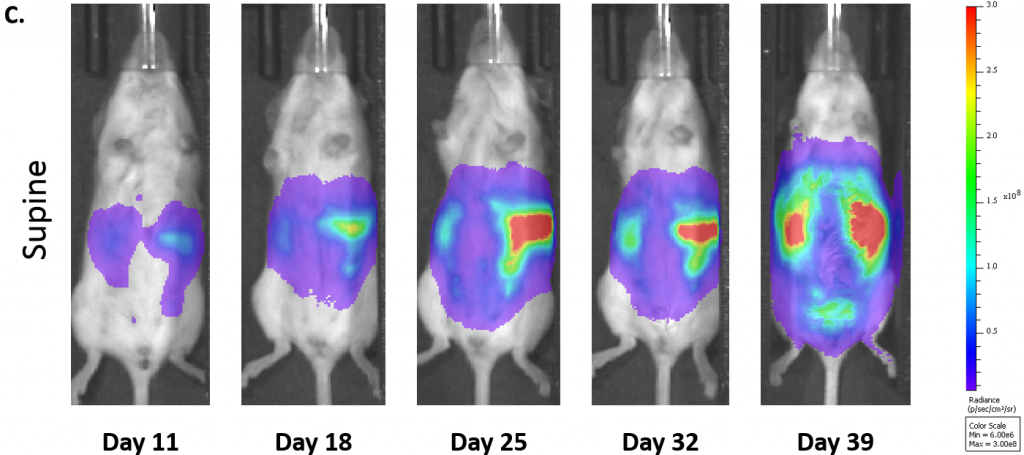
Fig. 1C : images représentatives par BLI de la progression du cancer de l'ovaire ID8-luc-mCh-Puro chez des souris albinos C57BL/6 (1,0×107 cellules/souris).
Les souris chez qui on a implanté des cellules ID8-Luc-mCh-Puro prennent du poids pendant la durée de l'étude (figure 2), certaines souris ne présentant une perte de poids qu'à la fin de l'étude. La progression de la maladie, qui se manifeste par une distension abdominale due à la formation d'ascites (figure 3), est linéaire avec une survie de 35 à 40 jours chez les souris albinos C57BL/6 implantées avec 1,0×107 cellules.
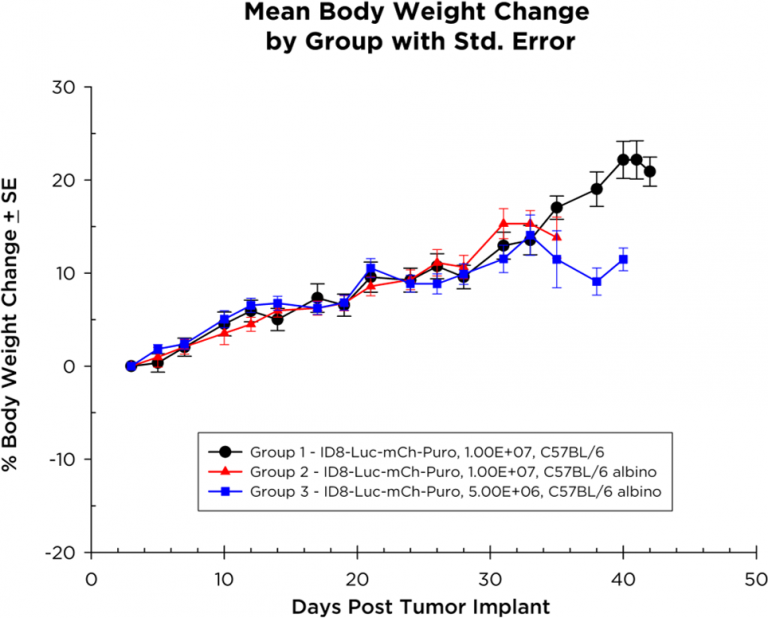
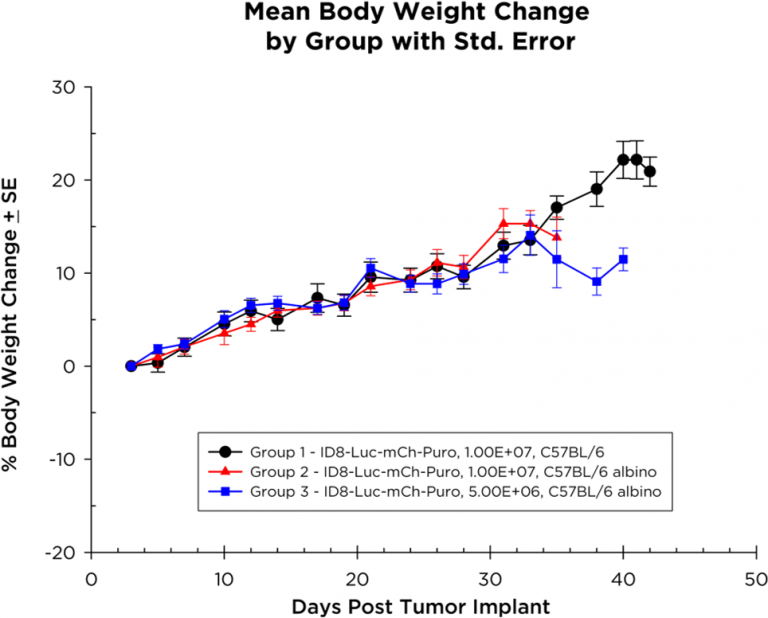
Fig. 3 : cinétique de survie des souris avec un cancer de l'ovaire ID8-luc-mCh-Puro orthotopique.
Labcorp performed immune profiling of the ascites from the ID8-Luc-mCh-Puro bearing mice. In Table 1, the immune profiles for total CD45+ leukocytes, CD4+ T cells, CD8+ T cells, Tregs, M-MDSC, G-MDSC, M1 TAM and M2 TAM are shown. While there is considerable heterogeneity in the immune profile between mice, all mice have a robust CD8+ T cell infiltration with favorable CD8+ T cell/Treg ratios. The ID8 ovarian cancer model has been reported to respond to IL-12 immunotherapy which combined with the favorable CD8+ T cell/Treg ratio suggests that the model should support testing of novel immunotherapies.2 Studies to analyze response to immunotherapies as well as standard of care are currently under development.
Tableau 1 : profilage par cytométrie en flux des sous-ensembles de cellules immunitaires dans des ascites murins individuels chez des souris avec cancer de l'ovaire ID8-Luc-mCh-Puro
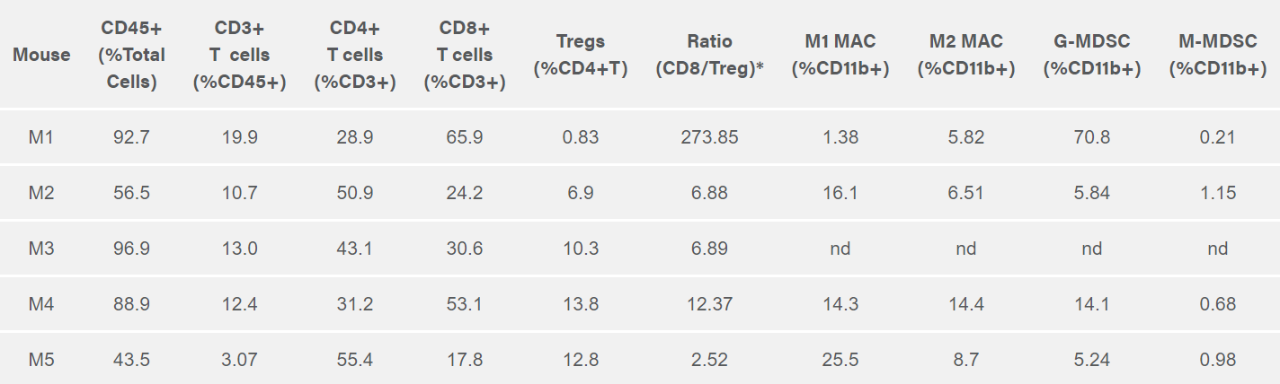
Tableau 1 : profilage par cytométrie en flux des sous-ensembles de cellules immunitaires dans des ascites murins individuels chez des souris avec cancer de l'ovaire ID8-Luc-mCh-Puro
*Le rapport CD8/Treg est déterminé à partir du comptage absolu des ensembles de CD8 et Treg individuels.
nd = non déterminé
Références
Connexion
Entamons le dialogue
Nous contacter